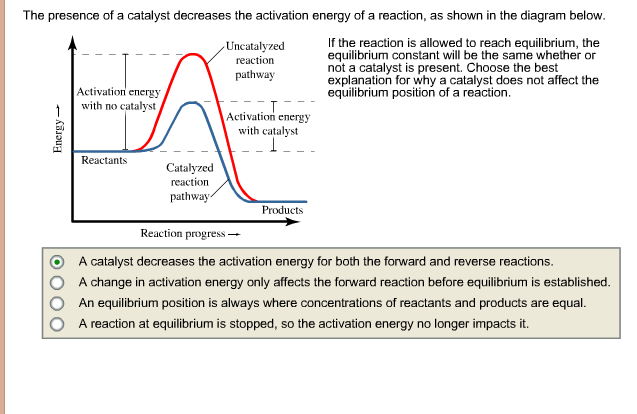
How much does catalyst need of activation energy

Rh dispersion values were calculated from the total H 2 chemisorption monolayer capacity.įrancesco Zaccaria.

Catalyst activation treatments were performed in situ on as-prepared supported Rh catalyst samples (0.5-0.7 g) using flowing UHP H 2 (~30 sccm) with an activation temperature/time sequence identical to that used for n-butane hydrogenolysis activity testing. H 2 chemisorption measurements were performed at room temperature (after evacuation to ~10 − 5 Pa) using an automated gas chemisorption unit (Coulter Omnisorp 100CX). After careful passivation, selected catalyst samples were characterized by x-ray diffraction and/or HREM. For all activity measurements, the reactor was assumed to operate in a differential mode. After 10 min of reactant flow over the catalyst at the desired reaction temperature, product gas analysis (relative to the pure n-butane reactant feed) was determined by on-line gas chromatography with a flame ionization detector. Reaction temperatures investigated ranged from 393 to 503 K, depending on the particular catalyst and its state of activation. 16, 17 Under steady state reactor conditions, UHP H 2 and research purity n-butane flow rates to the reactor were 20.0 and 1.0 sccm, respectively, and were selected to prevent catalyst deactivation. The model reaction used to determine the activity of the catalyst materials was n-butane hydrogenolysis, a known structure-sensitive reaction.
In certain cases, oxidative heat treatments at various temperatures utilizing He:O 2 gas mixtures (20:2 sccm He:O 2) were either performed prior to H 2 reduction or used as part of additional oxidation/reduction cycling experiments. For these activity studies, a single as-prepared catalyst sample (0.020-0.050 g) was activated in flowing UHP H 2 (20 sccm), and evaluated intermittently as a function of time (1–32 h) at a given activation temperature, with the activation temperature increased stepwise from 423 K, to 573 K, to 673 K. For all activation procedures and activity measurements, total gas pressure in the reactor was 630 mm Hg. The microflow reactor system was composed of a 0.635 cm OD quartz U-tube packed with quartz wool supporting the catalyst (fine powder form). Datye, in Studies in Surface Science and Catalysis, 1998 2.3 Catalyst Activation, Testing, and CharacterizationĬatalyst activation was performed in situ in a microflow reactor system prior to catalyst activity measurements.
